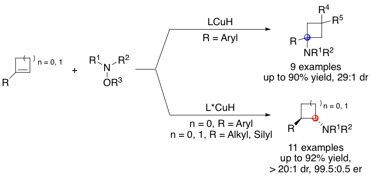
Amine-substituted cyclobutanes and cyclopropanes are important substructures in biologically active compounds. Moreover, many of the cycloalkane units bear multiple substituents and stereocenters. Therefore, synthetic methods that produce polysubstituted aminocyclobutanes and aminocyclopropanes in a highly diastereo- and enantioselective manner are of importance. Herein, we describe the diastereo- and enantioselective synthesis of various types of polysubstituted aminocyclobutanes and aminocyclopropanes through CuH-catalyzed hydroamination of 1-substituted cyclobutenes and cyclopropenes. These strained trisubstituted alkenes exhibit much higher reactivity compared to their unstrained analogues in the initial hydrocupration step of the reaction. Moreover, an interesting reversal of regioselectivity was observed in the hydroamination of 1-aryl-substituted cyclobutenes compared to the cyclopropene analogues. The origins of the enhanced reactivity of strained trisubstituted alkenes as well as the differences in the regio and enantioselectivity between reactions with cyclobutenes and cyclopropenes were investigated computationally.
