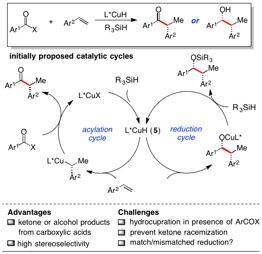
A new method for the enantioselective reductive coupling of aryl alkenes with activated carboxylic acid derivatives via copper hydride catalysis is described. Dual catalytic cycles are proposed, with a relatively fast enantioselective hydroacylation cycle followed by a slower diastereoselective ketone reduction cycle. Symmetrical aryl carboxyclic anhydrides provide access to enantioenriched α-substituted ketones or alcohols with excellent stereoselectivity and functional group tolerance.A new method for the enantioselective reductive coupling of aryl alkenes with activated carboxylic acid derivatives via copper hydride catalysis is described. Dual catalytic cycles are proposed, with a relatively fast enantioselective hydroacylation cycle followed by a slower diastereoselective ketone reduction cycle. Symmetrical aryl carboxyclic anhydrides provide access to enantioenriched α-substituted ketones or alcohols with excellent stereoselectivity and functional group tolerance.
