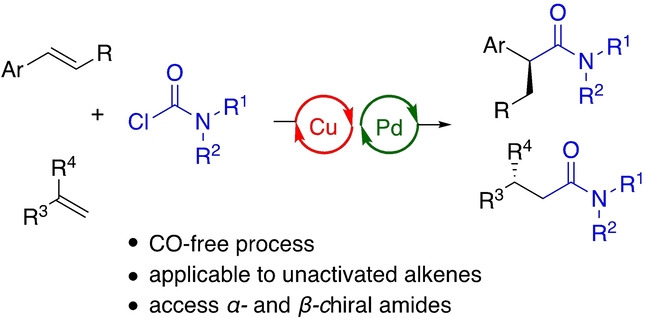
The asymmetric hydroaminocarbonylation of olefins represents a straightforward approach for the synthesis of enantioenriched amides, but is hampered by the necessity to employ CO gas, often at elevated pressures. We herein describe, as an alternative, an enantioselective hydrocarbamoylation of alkenes leveraging dual copper hydride and palladium catalysis to enable the use of readily available carbamoyl chlorides as a practical carbamoylating reagent. The protocol is applicable to various types of olefins, including alkenyl arenes, terminal alkenes, and 1,1-disubstituted alkenes. Substrates containing a diverse range of functional groups as well as heterocyclic substructures undergo functionalization to provide α - and β -chiral amides in good yields and with excellent enantioselectivities.
