A Neophyl Palladacycle as an Air- and Thermally Stable Precursor to Oxidative Addition Complexes
“A Neophyl Palladacycle as an Air- and Thermally Stable Precursor to Oxidative Addition Complexes”, Org. Lett., 2021, 23(20), 7927–7932.
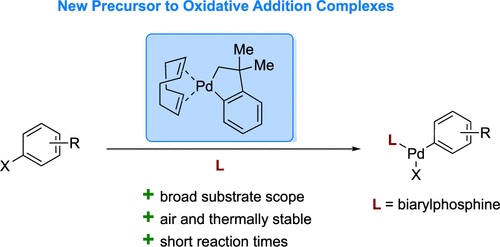
The utilization of isolated Palladium Oxidative Addition Complexes (OACs) has had a significant impact on Pd-catalyzed and Pd-mediated cross-coupling reactions. Despite their importance, widespread utility of OACs has been limited by the instability of their precursor complexes. Herein, we report the use of Cámpora’s palladacycle as a new, more stable precursor to Pd OACs. Using this palladacycle, a series of biarylphosphine ligated OACs, including those with pharmaceutical-derived aryl halides and relevance to bioconjugation, were prepared.