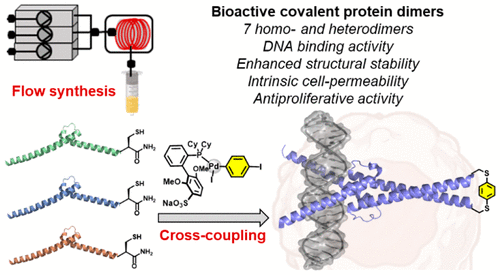
Transcription factors (TF), such as Myc, are proteins implicated in disease pathogenesis, with dysregulation of Myc expression in 50% of all human cancers. Still, targeting Myc remains a challenge due to the lack of small molecule binding pockets in the tertiary structure. Here, we report synthetic covalently linked TF mimetics that inhibit oncogenic Myc-driven transcription by antagonistic binding of the target DNA-binding site. We combined automated flow peptide chemistry with palladium(II) oxidative addition complexes (OACs) to engineer covalent protein dimers derived from the DNA-binding domains of Myc, Max, and Omomyc TF analogs. Palladium-mediated cross-coupling of synthesized protein monomers resulted in milligram quantities of seven different covalent homo- and heterodimers. The covalent helical dimers were found to bind DNA and exhibited improved thermal stability. Cell-based studies revealed the Max-Max covalent dimer is cell- penetrating and interfered with Myc-dependent gene transcription resulting in reduced cancer cell proliferation (EC50 of 6 μM in HeLa). RNA sequencing and gene analysis of extracted RNA from treated cancer cells confirmed that the covalent Max-Max homodimer interferes with Myc-dependent transcription. Flow chemistry, combined with palladium(II) OACs, has enabled a practical strategy to generate new bioactive compounds to inhibit tumor cell proliferation.
