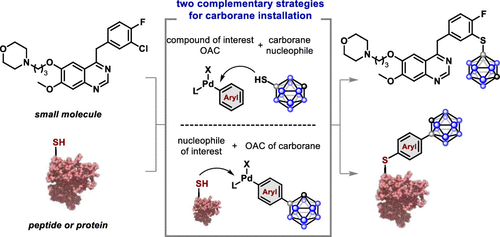
Carboranes represent a class of compounds with increasing therapeutic potential. However, few general approaches to readily embed carboranes into small molecules, peptides, and proteins are available. We report a strategy based on palladium-mediated C–X (X = C, S, and N) bond formation for the installation of carborane-containing moieties onto small molecules and peptides. We demonstrate the ability of Pd-based reagents with appropriate ligands to overcome the high hydrophobicity of the carborane group and enable chemoselective conjugation of cysteine residues at room temperature in aqueous buffer. Accordingly, carboranes can be efficiently installed on proteins by employing a combination of a bis-sulfonated biarylphosphine-ligated Pd reagent in an aqueous histidine buffer. This method is successfully employed on nanobodies, a fully synthetic affibody, and the antibody therapeutics trastuzumab and cetuximab. The conjugates of the affibody ZHER2 and the trastuzumab antibody retained binding to their target antigens. Conjugated proteins maintain their activity in cell-based functional assays in HER2-positive BT-474 cell lines. This approach enables the rapid incorporation of carborane moieties into small molecules, peptides, and proteins for further exploration in boron neutron capture therapy, which requires the targeted delivery of boron-dense groups.
